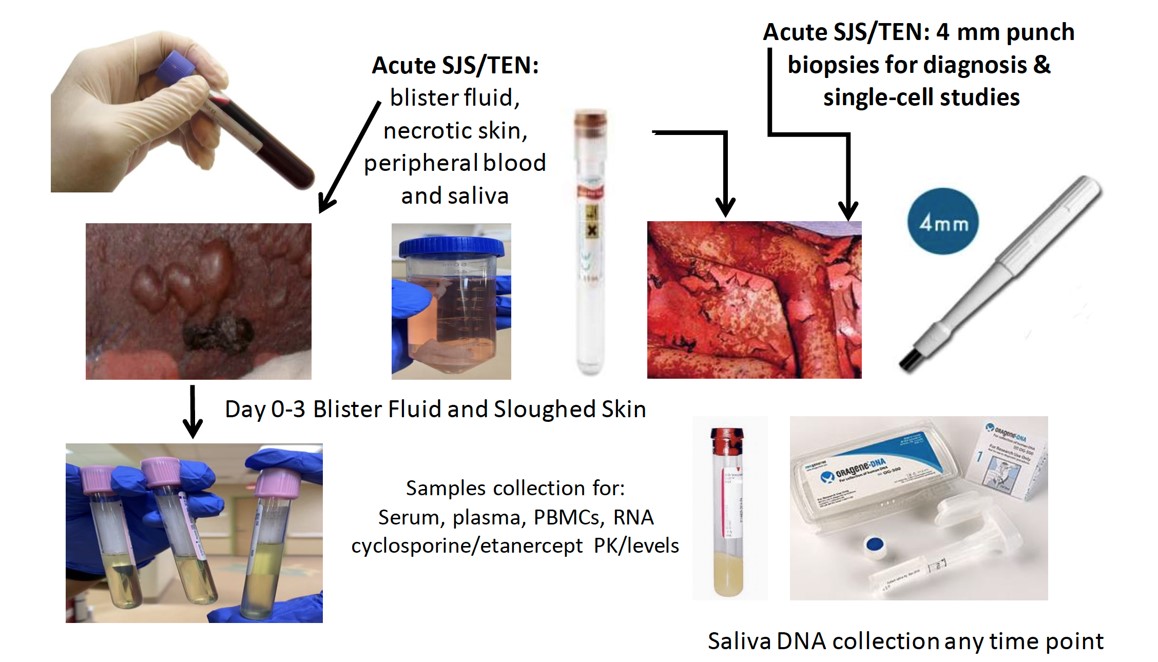
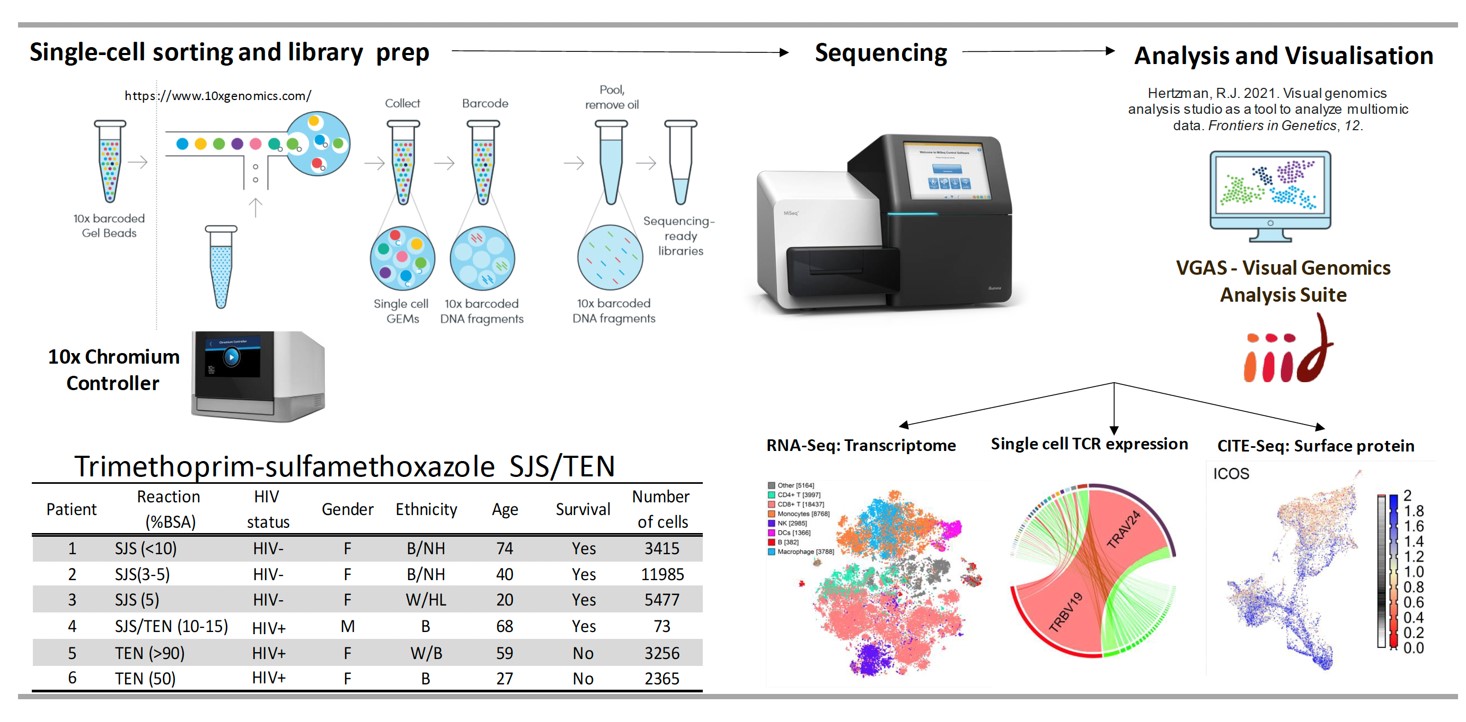
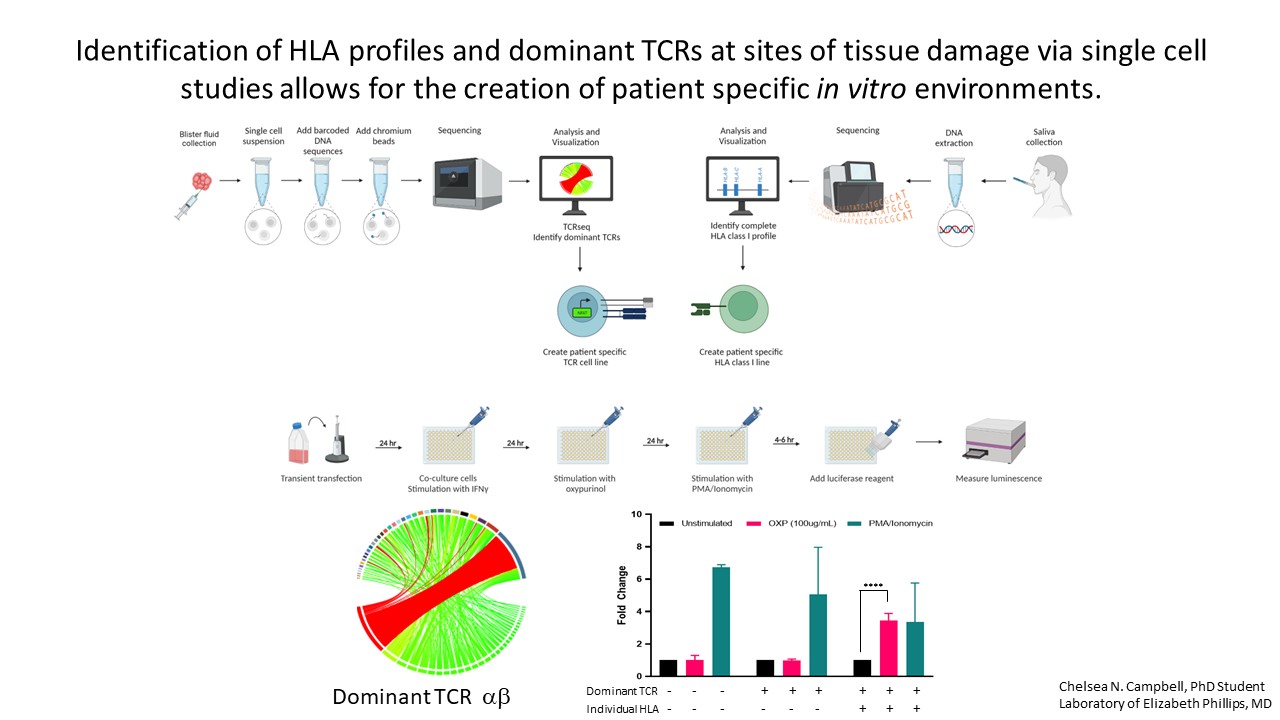
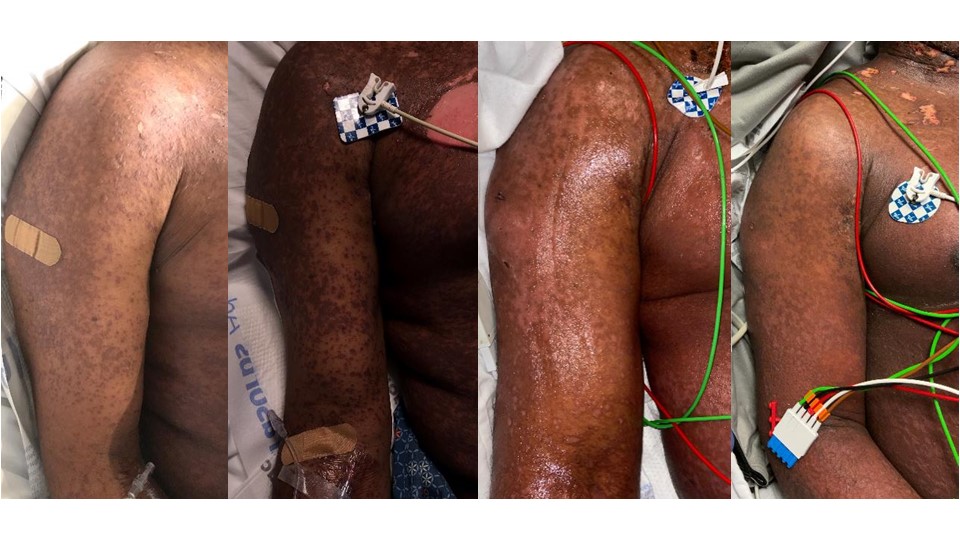
Defining HLA risk alleles associated with SCAR in a drug specific manner can improve prevention efforts and drug safety across patient populations. Successful pharmacogenomic screening is dependent upon the development of simple, robust, cost-effective testing that can be implemented in routine diagnostic laboratories.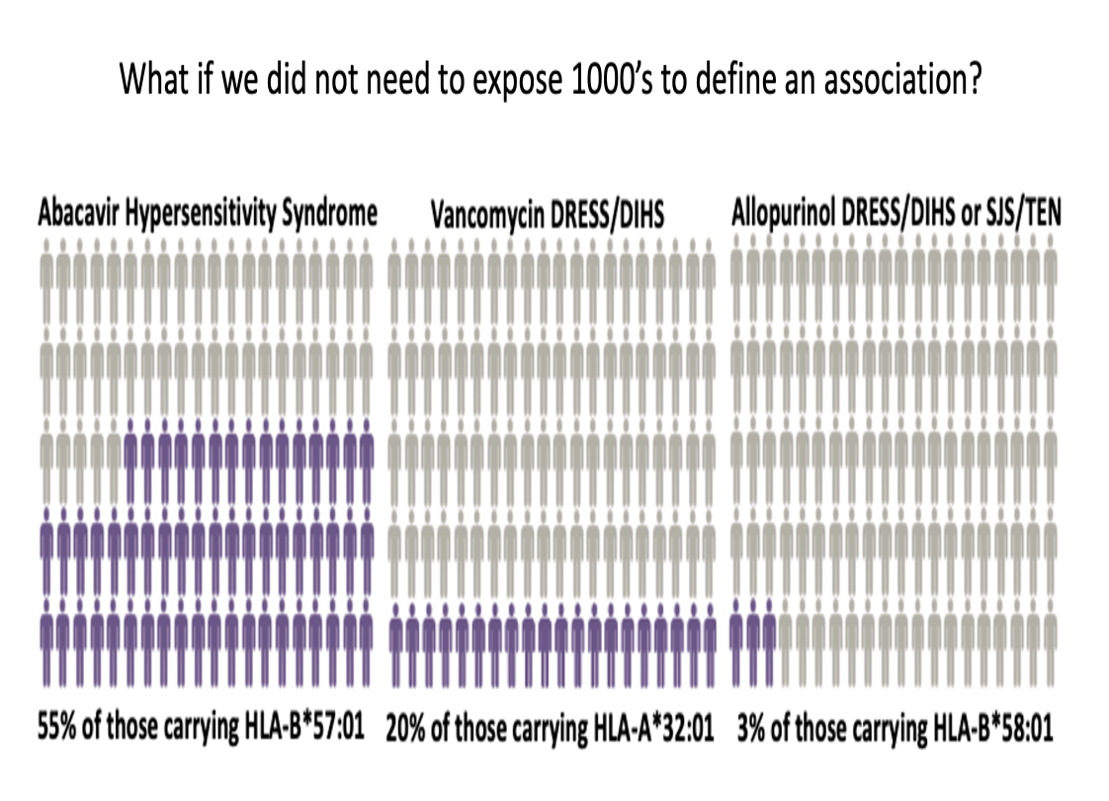
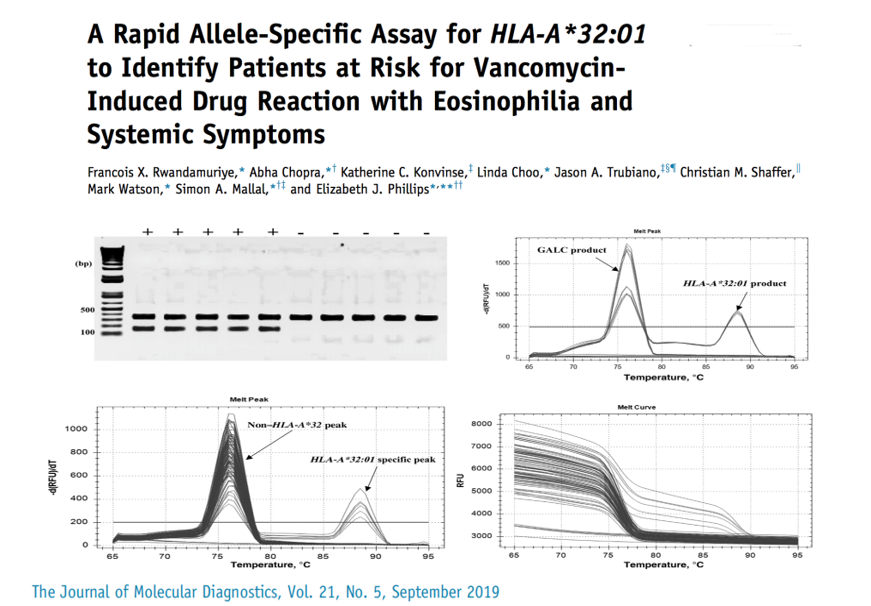
CDSI previously identified an association between the development of Vancomycin-induced DRESS and HLA-A*32:01. In order to identify patients at risk of developing an adverse reaction, a rapid allele test for HLA-A*32:01 was developed and can be used in the clinical setting as a pre-prescription assessment.
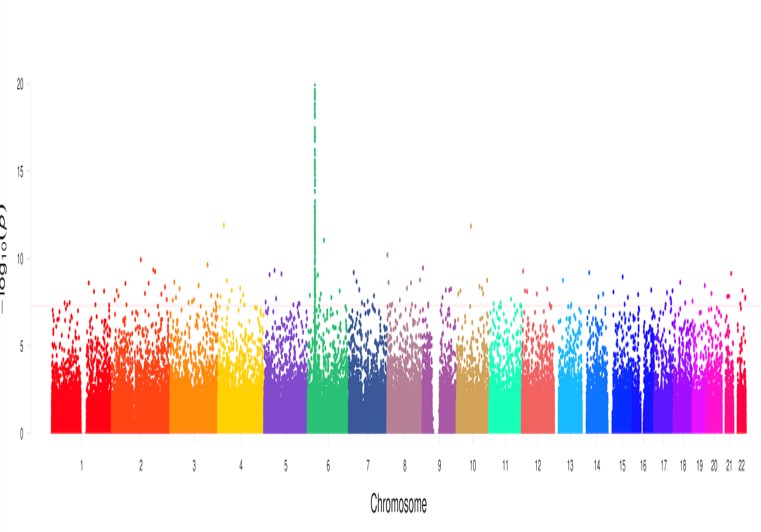
Discovery of Key Genetic Risk factors for Severe Drug-induced diseases such as Abacavir Hypersensitivity, Drug Reactions and Eosinophilia and Stevens-Johnson Syndrome and Toxic Epidermal Necrolysis